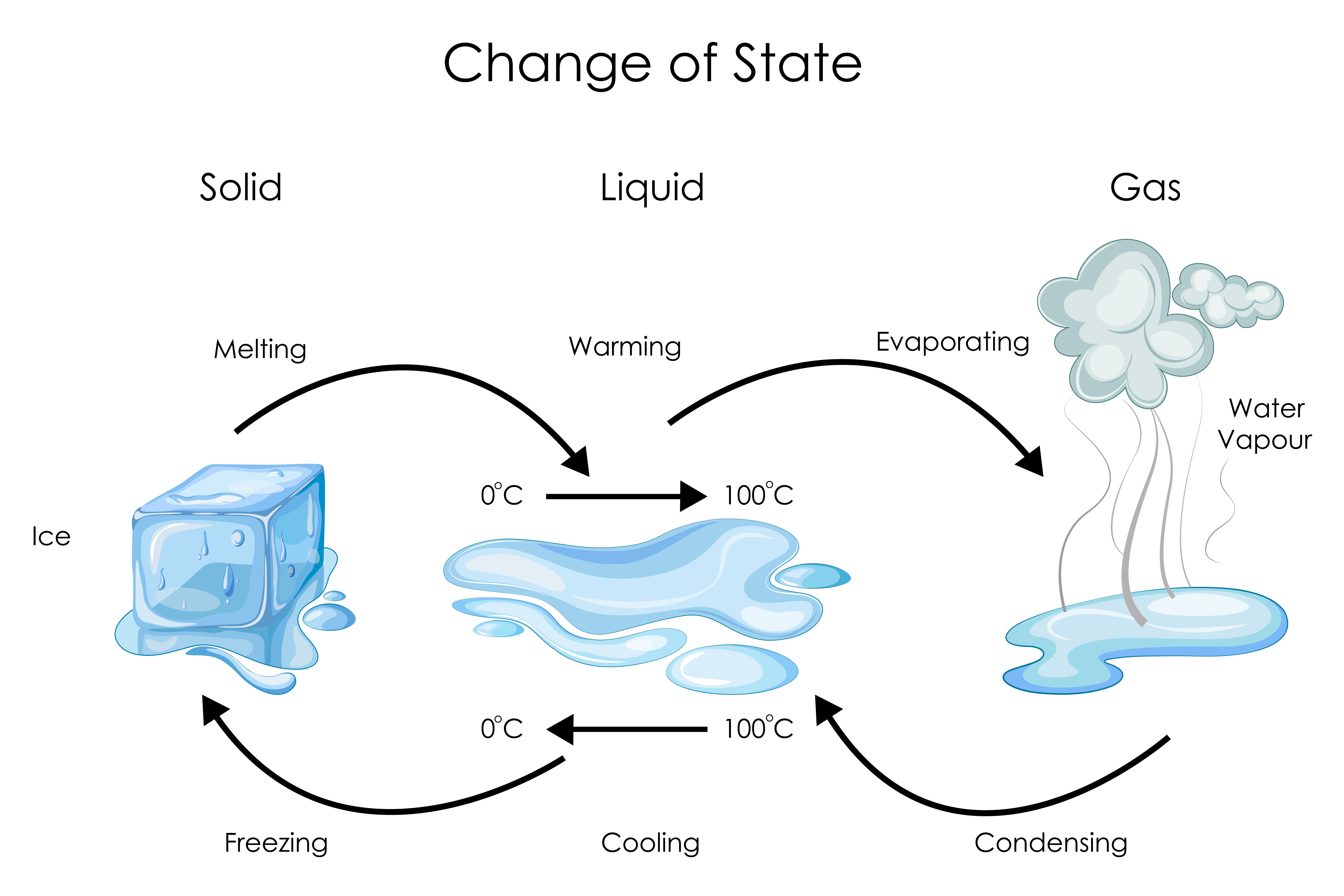
Itinerant Mission 3 Physical States of Matter Solid Liquid Gas
A typical phase diagram. The solid green line shows the behaviour of the melting point for most substances; the dotted green line shows the anomalous behavior of water. The red lines show the sublimation temperature and the blue line the boiling point, showing how they vary with pressure.
Illustration for Changes of State between Solid, Liquid and Gas Stock
Using the phase diagram for carbon dioxide provided, we can determine that the state of CO 2 at each temperature and pressure given are as follows: (a) liquid; (b) solid; (c) gas; (d) liquid; (e) gas; (f) gas. Exercise 10.5.2 10.5. 2. Identify the phase changes that carbon dioxide will undergo as its temperature is increased from −100 °C.
:max_bytes(150000):strip_icc()/phase-changes-56a12ddd3df78cf772682e07.png)
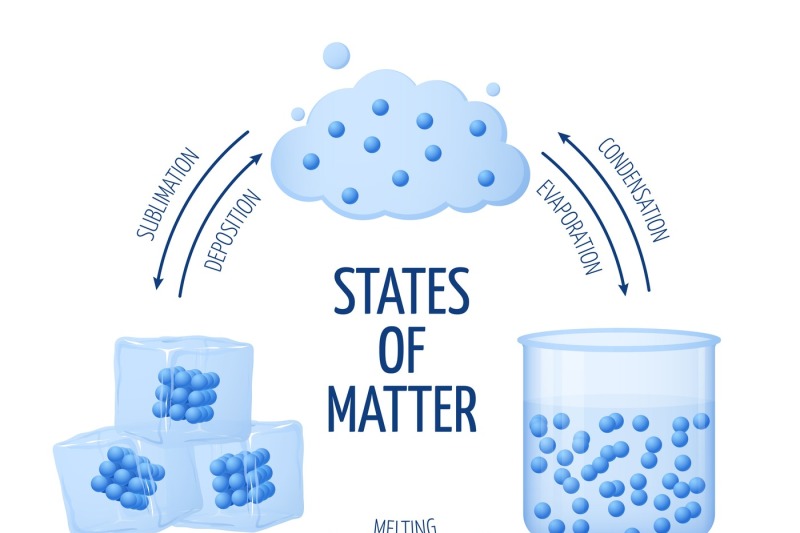
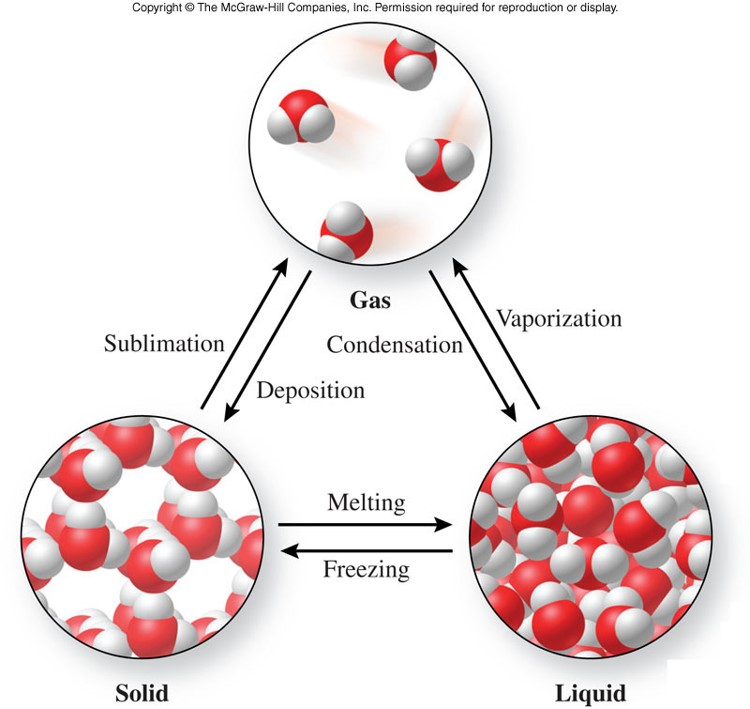
List of Phase Changes Between States of Matter
Watch different types of molecules form a solid, liquid, or gas. Add or remove heat and watch the phase change. Change the temperature or volume of a container and see a pressure-temperature diagram respond in real time. Relate the interaction potential to the forces between molecules.
Different states of matter solid, liquid, gas vector diagram By
Consider the phase diagram for carbon dioxide shown in Figure 5 as another example. The solid-liquid curve exhibits a positive slope, indicating that the melting point for CO 2 increases with pressure as it does for most substances (water being a notable exception as described previously). Notice that the triple point is well above 1 atm, indicating that carbon dioxide cannot exist as a liquid.
Chapter 1 Presentation
A phase diagram combines plots of pressure versus temperature for the liquid-gas, solid-liquid, and solid-gas phase-transition equilibria of a substance.
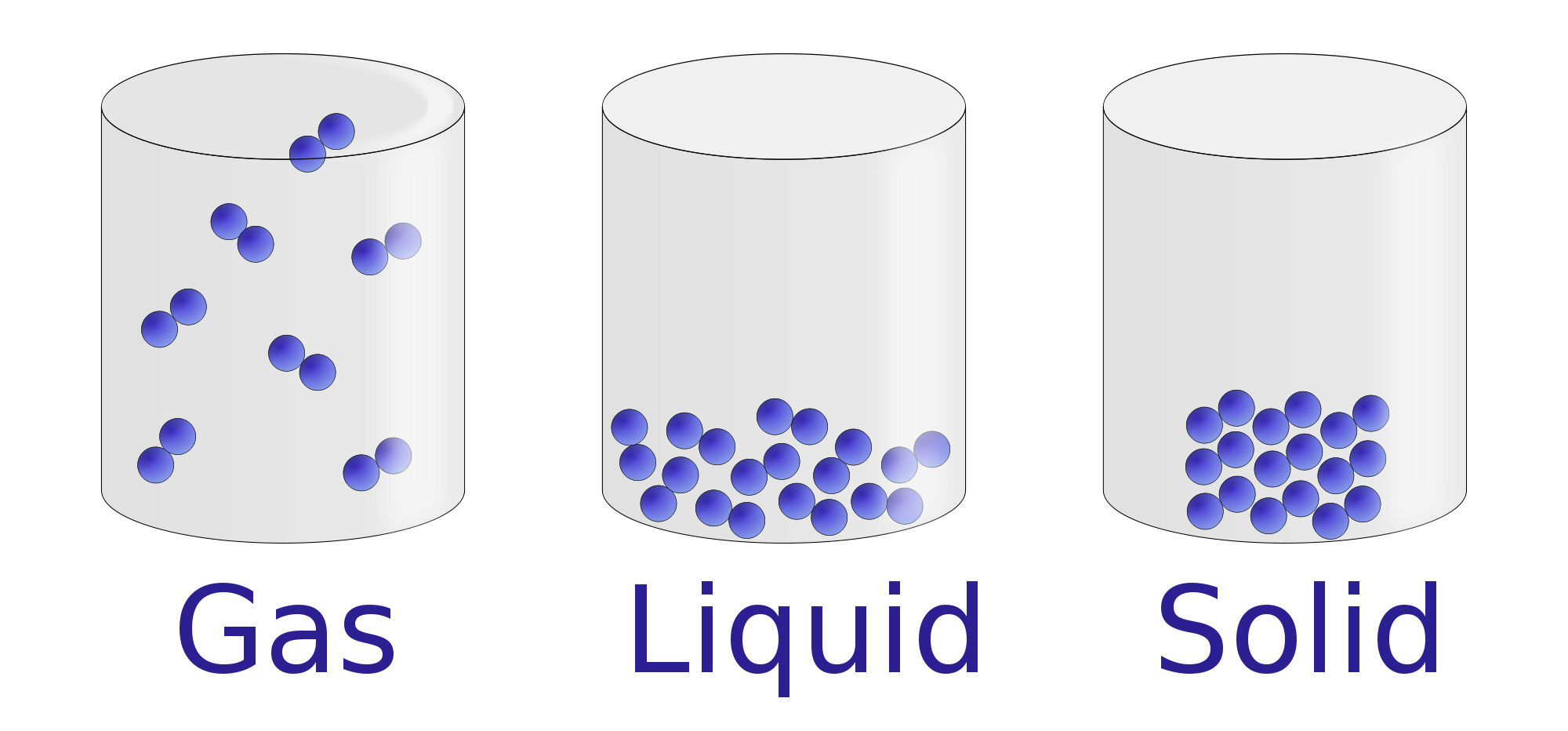
Properties of Liquids Chemistry Visionlearning
In the video here, Sal uses a horizontal line through the phase diagram. But, it doesn't have to be horizontal. Imagine a vertical line through this diagram-- for water, choose 100 degrees C. As long as you are at 100 C, you can change the phase by changing the pressure on the system.
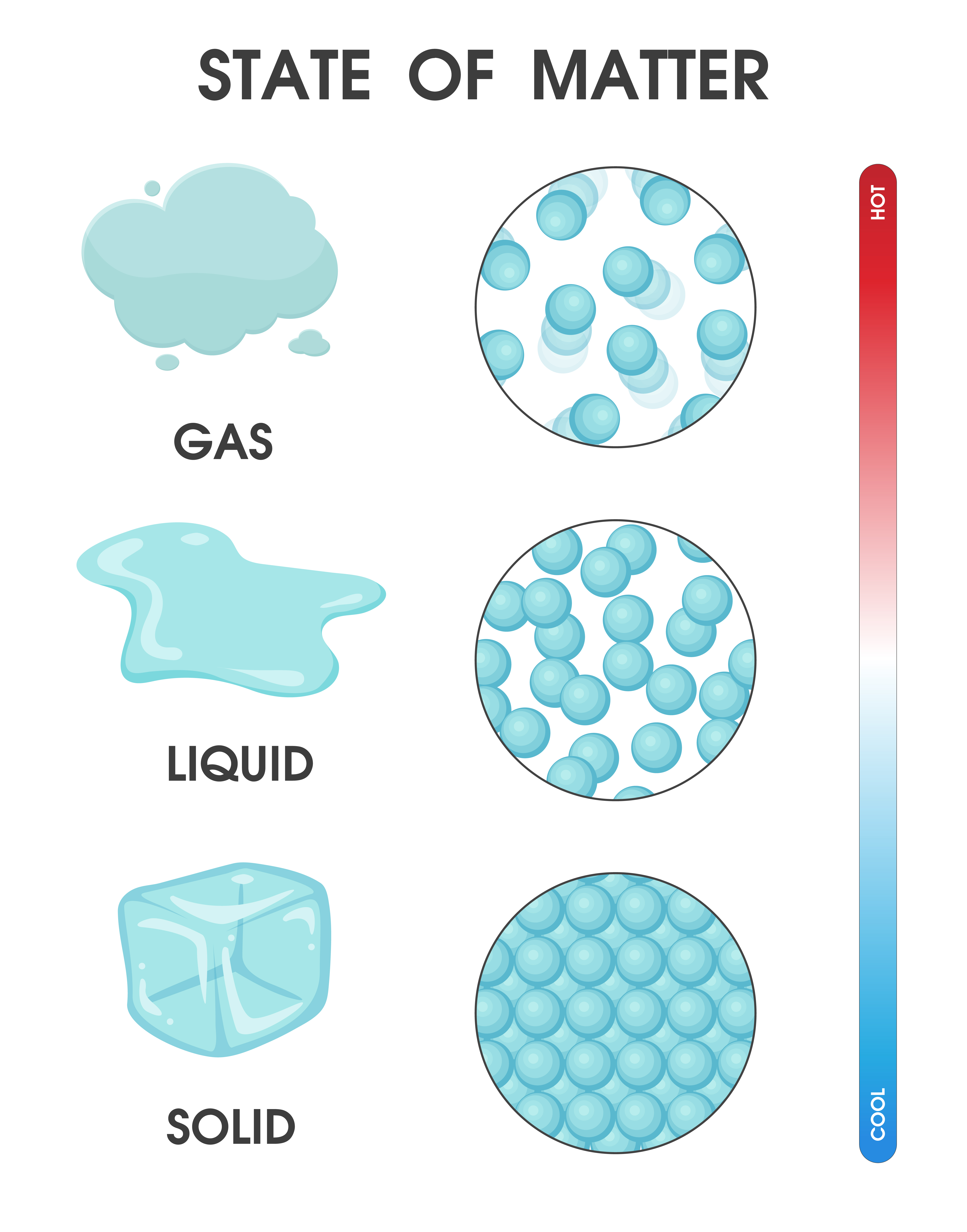
Changing the state of matter from solid, liquid and gas due to
Solids Liquids Gases The particle model What are models? Working scientifically Variables Test your knowledge Key points Almost everything is made of particles. Particles can be atoms,.
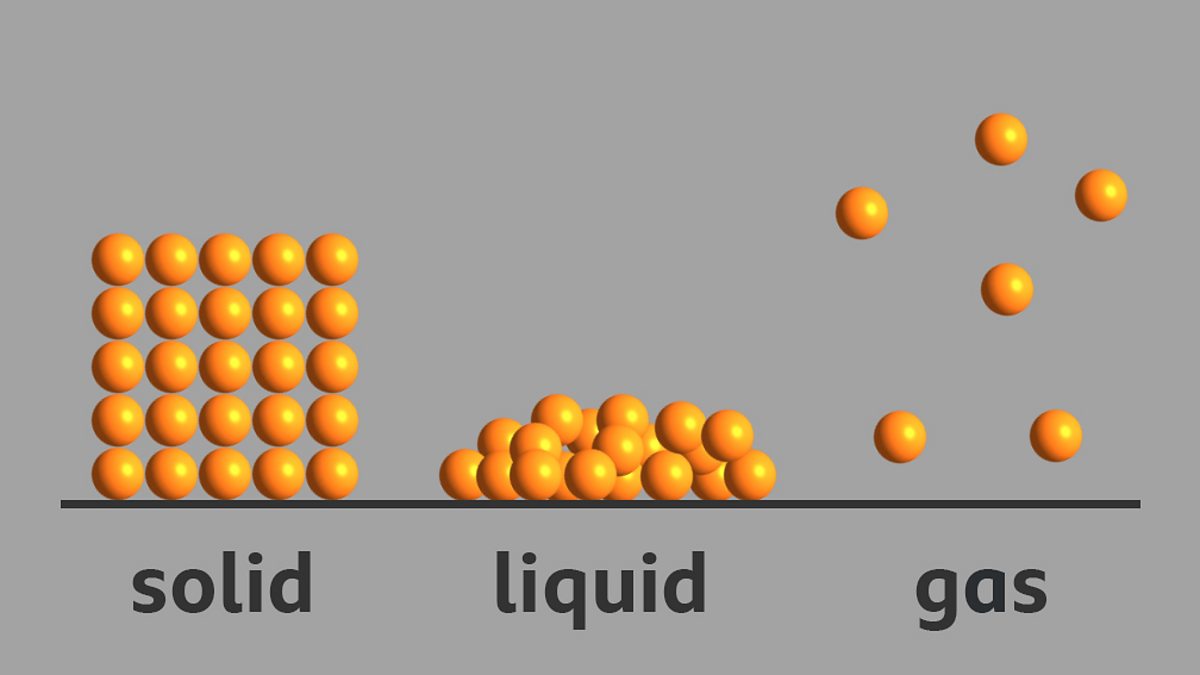
What is the arrangement of particles in a solid, liquid and gas? BBC
A phase diagram lets you work out exactly what phases are present at any given temperature and pressure. In the cases we'll be looking at on this page, the phases will simply be the solid, liquid or vapor (gas) states of a pure substance. This is the phase diagram for a typical pure substance. These diagrams (including this one) are nearly.
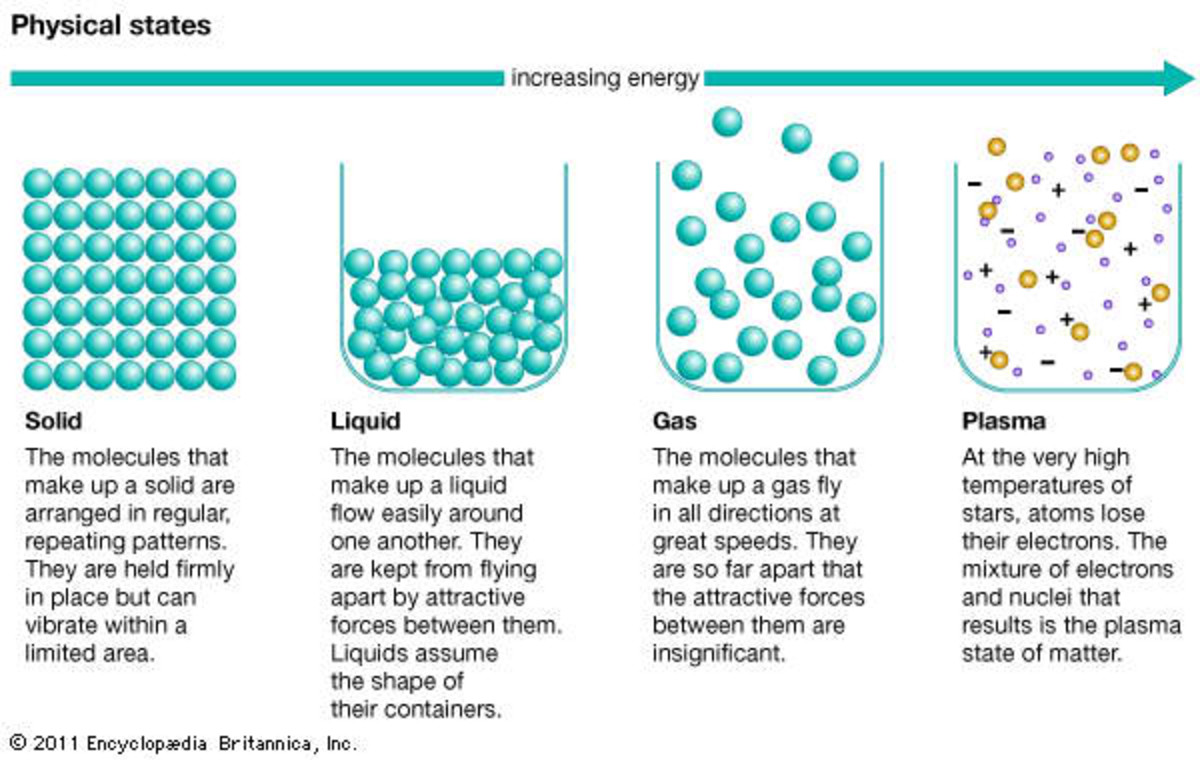
All About Matter An Introduction to the Basics Owlcation
GCSE AQA The three states of matter - AQA Solids, liquids and gases The three states of matter can be represented by the particle model. This model explains the properties of substances in their.

IGCSE Edexcel Chemistry Help 1.1 understand the arrangement, movement
Gas. The atoms and molecules in gases are much more spread out than in solids or liquids. They vibrate and move freely at high speeds. A gas will fill any container, but if the container is not sealed, the gas will escape. Gas can be compressed much more easily than a liquid or solid.

KS2 Science Year 4 3 States of Matter Solids Liquids and Gases
A phase diagram combines plots of pressure versus temperature for the liquid-gas, solid-liquid, and solid-gas phase-transition equilibria of a substance. These diagrams indicate the physical states that exist under specific conditions of pressure and temperature, and also provide the pressure dependence of the phase-transition temperatures (melting points, sublimation points, boiling points).
Chemistry States Of Matter Level 2 activity for kids PrimaryLeap.co.uk
A phase transition occurs when a substance changes from one state of matter to another state. There are three primary states of matter: liquid, solid, and gas. A phase diagram is a plot that illustrates the different phases of a substance across multiple variables, most often temperature and pressure. The diagram can help to demonstrate how.

Solids, liquids and gases
12.4: Phase Diagrams. To understand the basics of a one-component phase diagram as a function of temperature and pressure in a closed system. To be able to identify the triple point, the critical point, and four regions: solid, liquid, gas, and a supercritical fluid. The state exhibited by a given sample of matter depends on the identity.
8.2 Solids and Liquids The Basics of General, Organic, and Biological
The intersection with the logarithmic curve for the gas will define an equilibrium pressure for gas-solid co-existence. Generally vapor pressures above solids are quite small, but not negligible. As for liquids we can construct a line representing the equilibrium pressures for sublimation as function of temperature and add it to the phase diagram.

Pin de tintinwin en nilar Estados de agregación de la materia
The solid-liquid curve labeled BD shows the temperatures and pressures at which ice and liquid water are in equilibrium, representing the melting/freezing points for water. Note that this curve exhibits a slight negative slope (greatly exaggerated for clarity), indicating that the melting point for water decreases slightly as pressure increases.

States of matter A simple introduction to solids, liquids, gases
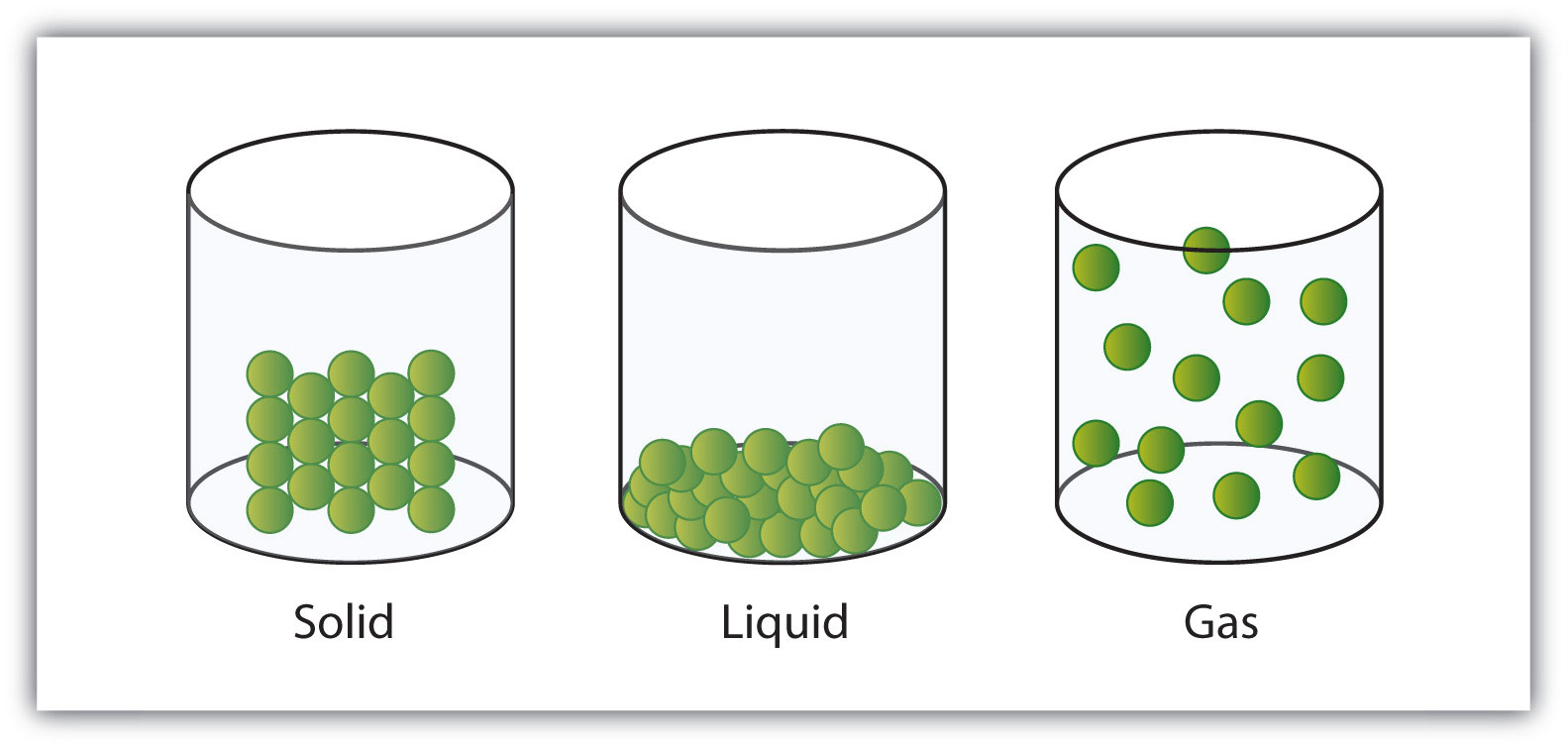
A solid is a state of matter in which atoms or molecules do not have enough energy to move. They are constantly in contact and in fixed positions relative to one another. Figure 6.1. 1: States of Matter. All three containers contain a substance with the same mass, but the substances are in different states.